 Namely, for a given macrostate of a system, its entropy is given by k Bln(Ω), where k B is called the Boltzmann constant, which is equal to 1.38*10 -23 Joules per Kelvin in SI units, and Ω represents the number of possible microstates of the particles that together would comprise the same macrostate – a concept which relies on the indistinguishability of certain configurations of identical particles. In statistical mechanics, the entropy of a system is defined in terms of the statistics of the motions of its constituent particles. From this view, entropy represents the amount of energy per unit temperature of a system that is unavailable to do useful work. Definitions of the properties of thermodynamic systems were also presumed to be in equilibrium, and were only much later extended to non-equilibrium situations. In classical thermodynamics, the details of a system’s constituents are not considered, but are instead described by their macroscopically averaged behavior. Note that although the loose definition of entropy as a measure of disorder suffices for the purposes of this discussion, it is not strictly correct. So, if the entropy of a system decreases (becomes more ordered), then that will be compensated for by an increase in the entropy (or disorder) of the surroundings, and vice versa. Energy is still conserved, but over time, less of it is available to do work. “No process is possible whose sole result is the absorption of heat from a reservoir and the conversion of this heat into work.”įor the purposes of this section, however, the 2 nd Law can be thought of as stating that the total disorder of the universe ( entropy) tends to increase over time. Whereas the Plank-Kelvin formulation of the 2 nd Law is stated as follows: “No process is possible whose sole result is the transfer of heat from a cooler to a hotter body,” The 2 nd Law is a little subtler, and can be stated and explained in different (albeit ultimately equivalent) ways, either in terms of classical or statistical thermodynamics.įor example, the Clausius statement of the 2 nd Law is that *As an interesting (albeit admittedly tangential) side-note, energy-momentum conservation is not so well-defined for certain cosmological applications of general relativity. An increase in a system’s internal energy requires an input of heat and/or work from the surrounding environment, whereas a decrease requires a release of heat from the system to the environment, and/or work done by the system on the environment. 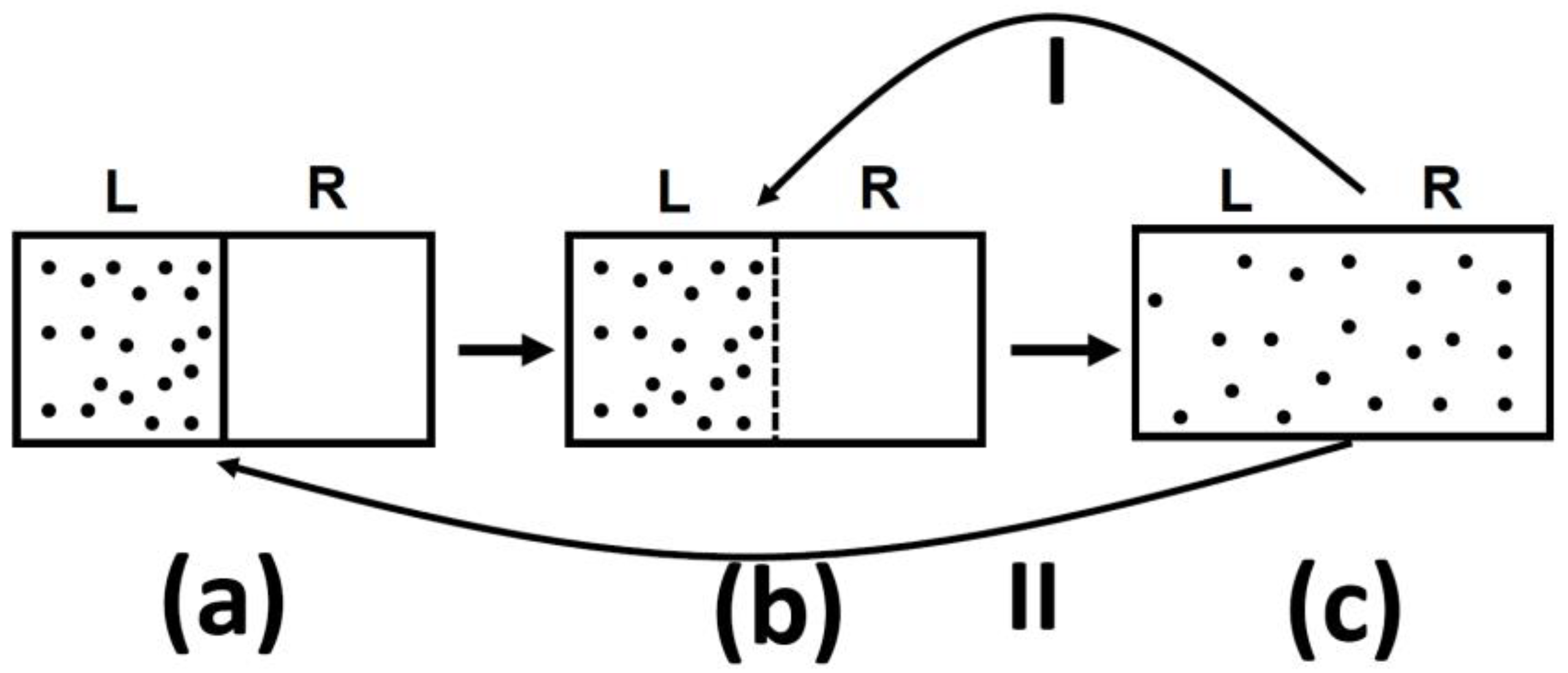
This means that changes in the internal energy of a thermodynamic system are compensated for by the surroundings such that the total energy of the universe is conserved. It is often conveyed by the statement that energy can be neither created nor destroyed in an isolated system, but rather can only change from one form of energy to another. The 1 st Law is just the law of conservation of energy, which states that the total energy of an isolated system is constant.  
In this way, scientists and engineers can choose to define the boundaries between the environment and the system they are analyzing in a way that makes it convenient to apply the 1 st and 2 nd Laws of Thermodynamics to them both. One of the fundamental concepts of thermodynamics is the idea of dividing the universe into a system + its surroundings. System vs surroundings and the Laws of Thermodynamics Examples of path dependent thermodynamic variables include heat and work. Examples of thermodynamic state functions include temperature, pressure, internal energy, density, entropy, and enthalpy. The displacement is therefore a state function, whereas the path length (and gas required) depends on the path taken. However, some of those paths would be less direct than others, and would therefore require you to travel a greater path length (and spend more on gas if you’re driving). On the other hand, path functions can take on very different values depending on the path by which the system arrived at a state from its previous state.įor example, there are probably a few different ways you could get from your house to your friend’s house across town, all of which would result in the same displacement between your home and your friend’s. They depend only on the starting and ending states of the system. State functions are properties of a system whose values do not depend on how they were arrived at from a prior state of the system. 
In thermodynamics, scientists distinguish between what are called state functions vs path functions. State Functions vs Path Dependent Functions 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
